Back in the 1980’s, after multiple lawsuits against vaccine manufacturers began causing unsustainable financial losses, Congress set up a system of “no liability” for drug manufacturers in exchange for an “excise tax” of 75 cents on every vaccine dose administered. The money collected is used to fund the Vaccine Injury Compensation Trust Fund, (a.k.a. “vaccine court”). The fund can provide compensation for individuals who sustain an adverse reaction to any FDA approved vaccine. The Department of Treasury collects the taxes and manages the Fund’s investments and produces Vaccine Injury Compensation Trust Fund Monthly Reports.
Comirnaty is the name of the newly licensed FDA approved version of the Pfizer COVID vaccine. Under Emergency Use Authorization, Pfizer enjoyed complete immunity from lawsuits and did not have to collect or pay the 75 cents tax on every dose administered. Now that Pfizer’s product has received full FDA approval, the licensed version falls under the same tax requirements set forth by the Vaccine Injury Compensation Trust Fund just like any other FDA licensed vaccine.
While FDA approval does not allow individuals to directly sue Pfizer for damages caused by an adverse reaction to the vaccine, individuals can apply for damages and compensation from the Vaccine Injury Trust Fund for harm caused by an adverse reaction from any FDA licensed vaccine.
A recently created program called the “Countermeasures Injury Compensation Fund”, was passed by Congress to provide increased “liability protections for pandemic and epidemic products and security countermeasures” specifically granted under EUA. Despite the promising name, the Countermeasures Injury Compensation fund merely strengthens legal protections for the manufacturers while also providing minimal “reimbursement for COVID-19 testing and treatment of uninsured individuals“. Unlike the Vaccine Injury Compensation program, claims filed under the CIC are limited to medical expenses directly incurred from COVID treatments or adverse reactions to EUA products, and only uninsured individuals are entitled to file a claim.
The number of adverse reactions reported to the Vaccine Adverse Event Reporting System (VAERS) from all currently administered COVID vaccines is nearing the 700,000 mark, a number that is certain to surpass all other vaccines combined.
Based on current VAERS data projections, the number of claims to the Vaccine Injury Compensation Trust Fund from Pfizer’s FDA approved Comirnaty vaccine will quickly bankrupt the fund. Bad press from the mounting claims will harm Pfizer’s ability to continue marketing the vaccine and/or the potential marketability of any new products designed to address the growing number of new COVID variants.
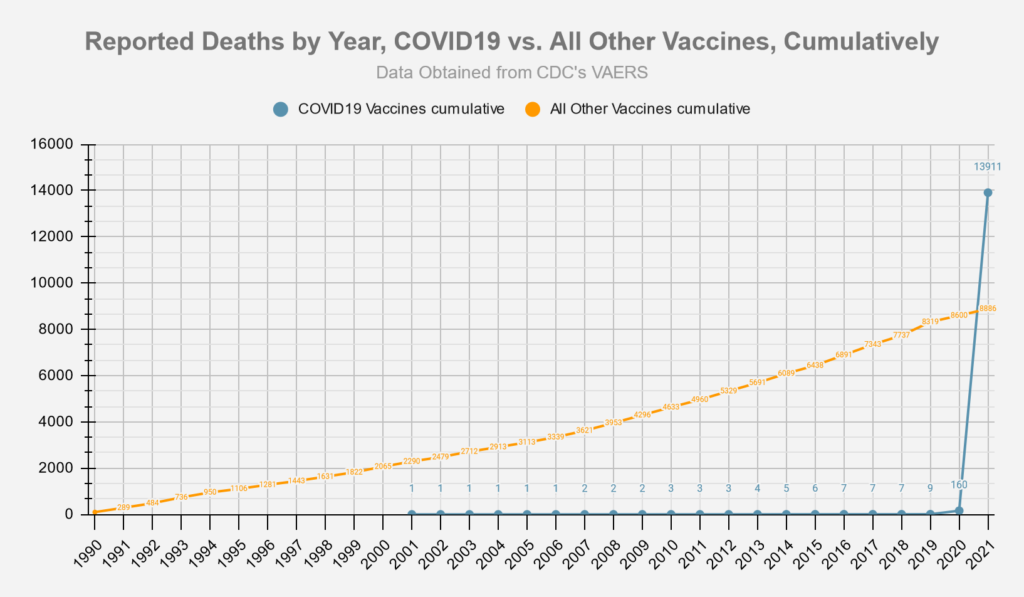
According to former Justice Department attorney Richard Topping, (currently the chief legal officer of CareSource Management Group Inc., a Dayton, Ohio-based non-profit that’s one of the nation’s largest Medicaid-managed health care plans), the number of claims expected to flood into the compensation program would quickly overwhelm the current system.
Topping has said that, “With potentially hundreds of millions of people getting vaccinated against one disease, even a very small percentage can translate into a lot of injured people.” The potential to quickly exhaust the Vaccine Injury Compensation Fund presents a real problem for Pfizer and other COVID vaccine manufacturers still seeking FDA approval.
It is vitally important to note that any current mandate by state governments, such as those being dictated by the Governor of Washington State, are relying on the fact that the vast majority of people are ignorant about the legal differences between a vaccine administered under EUA and one administered under full FDA license. Even pharmacists are unaware that current Pfizer vaccine stocks are still under EUA and are not FDA licensed.
According to the FDA license, the vials must say, “Comirnaty” and have the FDA license number in order for the vaccine to qualify for inclusion in the Vaccine Injury Compensation Fund. Again, (with the very limited CIC exception noted above), it is important to note that injuries sustained from vaccines administered under EUA do not qualify for compensation!
Needless to say, Pfizer is not happy with the current FDA requirements under the licensing agreement. The company is seeking the same legal immunity for their licensed product that it has enjoyed under EUA authorization. While the formulations are the same, there are distinct legal differences between the products. People have a right to know that when they receive the current EUA authorized version of the Pfizer COVID vaccine, they are not eligible to submit a claim for injuries. This is typically known in the con game as “bait and switch”.
Sources for this article include:
https://www.fda.gov/media/151710/download (FDA letter approving Pfizer vaccine)
https://www.insurancejournal.com/news/national/2020/08/14/579150.htm

Listen to Dr. Malone, the inventor of mRNA technology, as he discusses the medical data and the potential risks of the vaccine.
https://rumble.com/vipvwl-pfizer-data-reveal-where-do-the-lipid-nanoparticles-collect-robert-malone-s.html